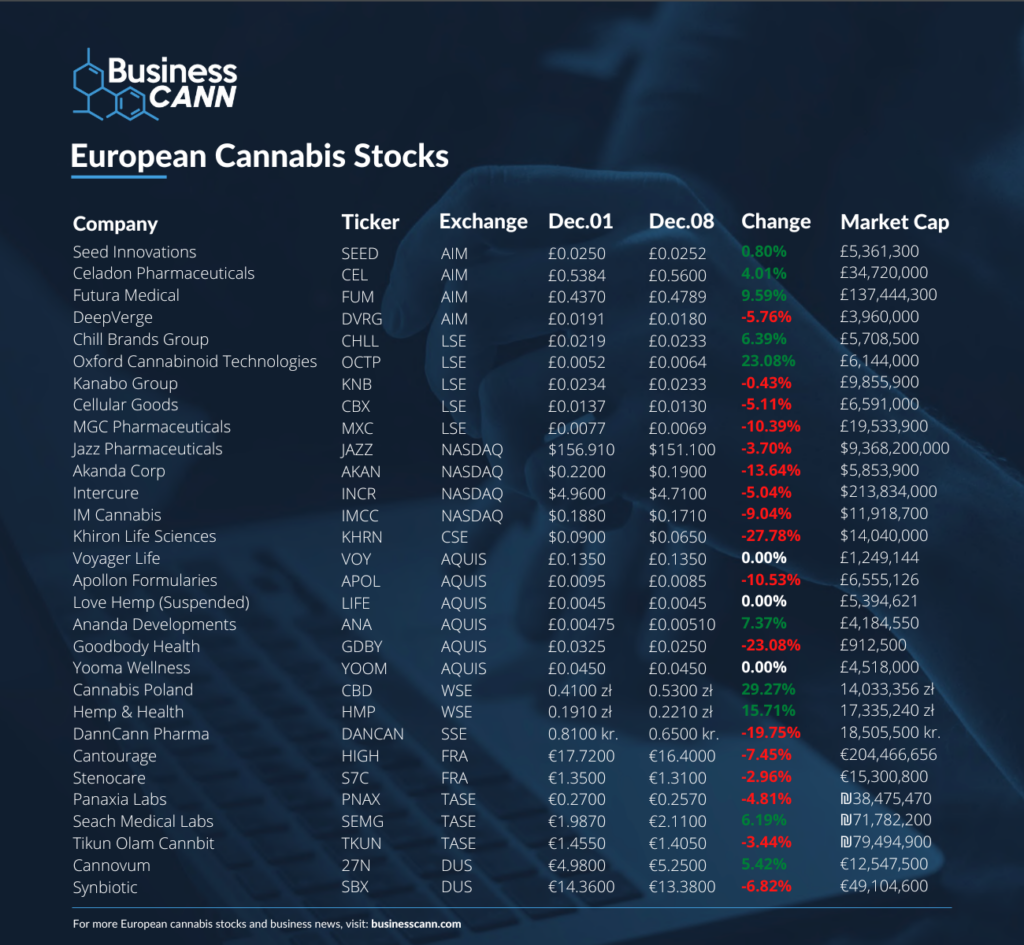
Oxford Cannabinoid Technologies
Oxford Cannabinoid Technologies (OCT) has seen its share price jump by more than 20% this week after announcing that its CEO Dr John Lucas would be stepping down.
The pharmaceutical company announced on Monday, December 5 that Dr Lucas would leave the company by the end of the year, staying on until then to ‘assist’ the company and facilitate a ‘smooth handover’.
In his place, OCT’s COO Clarissa Sowemimo-Coker will take over as interim CEO, and the company plans to recruit a General Counsel and Company Secretary to take on her previous responsibilities.
According to the company, Ms Sowemimo-Coker will act as CEO ‘while the company completes its Phase I clinical trials’ for its flagship compound, which are due to commence in January 2023 with results due in early Q2 2023.
The company made no indication regarding if or when it planned to hire a new permanent CEO. However, when asked whether she expected to still be in the position when the trials were complete, Ms Sowemimo-Coker told Proactive Investors: “With a bit of luck, I’m sure I will be. The plan is for me to be taking the reins as we get through that very, very important period.”
Dr Lucas’ departure will be the second major change to the company’s boardroom in as many months, following the appointment of William Smalley as its new group finance director in October.
During his tenure, Dr Lucas saw OCT become one of the first cannabis companies in history to launch on the London Stock Exchange, securing numerous high-profile investors, including Imperial Brands and Snoop Dogg’s cannabis vehicle Casa Verde Capital.
Since its IPO, OCT has seen a near 90% drop in its stock price, forcing it to delay the start of trials for its other lead compounds.
The company has also weathered a number of controversies over the past year, culminating in a shareholder vote over whether to oust the entire board, which ultimately came to nothing.
Dr Lucas: “I have very much enjoyed my tenure at OCTP. It has been a challenging but rewarding experience.
“However, I feel the time is now right to step aside. I’m leaving the Company in very capable hands and will be closely following its continued development. I wish the team every success in the future.”
Investors have responded positively to the directorate change, with many seeing it as a clean break for the company.
Greencare Capital
Greencare Capital, an Aquis-listed investment vehicle focused on businesses in CBD, wellness and hemp, has announced its plans to move out of the cannabis sector entirely and change its name.
In an RNS to investors published yesterday (December 8), Greencare said that its board of directors had concluded that ‘it is in the best interests of all shareholders for the company to withdraw from the medicinal cannabis, CBD and related wellness markets’.
Should shareholders vote to approve its new strategy in a General Meeting on December 28, 2022, Greencare would change its name to MaxRS Ventures and begin targeting ‘distressed or out-of-favour assets’ in the life sciences, crypto, retail and environmental technology sectors.
According to Greencare, its exit from the cannabis space is largely down to the current state of CBD regulation and the slow progress of the novel foods legislation across the UK and Europe.
During its annual report for the year ended October 2021, Greencare’s directors warned that novel foods regulation would ‘likely cause short to medium term turbulence in the market’, and that legislation was still to be agreed in Europe, meaning investments in the space have ‘proved difficult’.
“During the period since this statement, the cannabis and related business sectors have generally continued to perform poorly and the sectors have not generated the quality or underlying value of investment opportunities originally envisaged by the company at the time of flotation in December 2019.”
Greencare has made relatively few investments during its tenure in the cannabis industry, with a portfolio consisting only of Clear CBD and Voyager Life, which it invested £100k into last year.
MGC Pharmaceuticals
MGC Pharmaceuticals announced during the week that it had now completed the $1m delivery of its flagship COVID drug Artemic to US distribution partner AMC Holdings.
In October, MGC’s deal with AMC was amended ‘owing to increasing demand’, seeing all 50,000 units delivered in a single shipment, marking the ‘first substantial commercial delivery of the product’ under the agreement.
According to AMC, the product will soon be available online and through independent pharmacies, offering customers a cost-effective COVID treatment now that the government has stopped supporting many patients.
Days later, MGC announced that preclinical studies had shown its other flagship drug, CimetrA, had demonstrated anti-inflammatory effects and could also show promise in treating the effects of COVID.
The studies’ results will reportedly form part of the US FDA application to register the drug as an Investigational New Drug in the US, a ‘critical milestone in the process of drug registration’.























