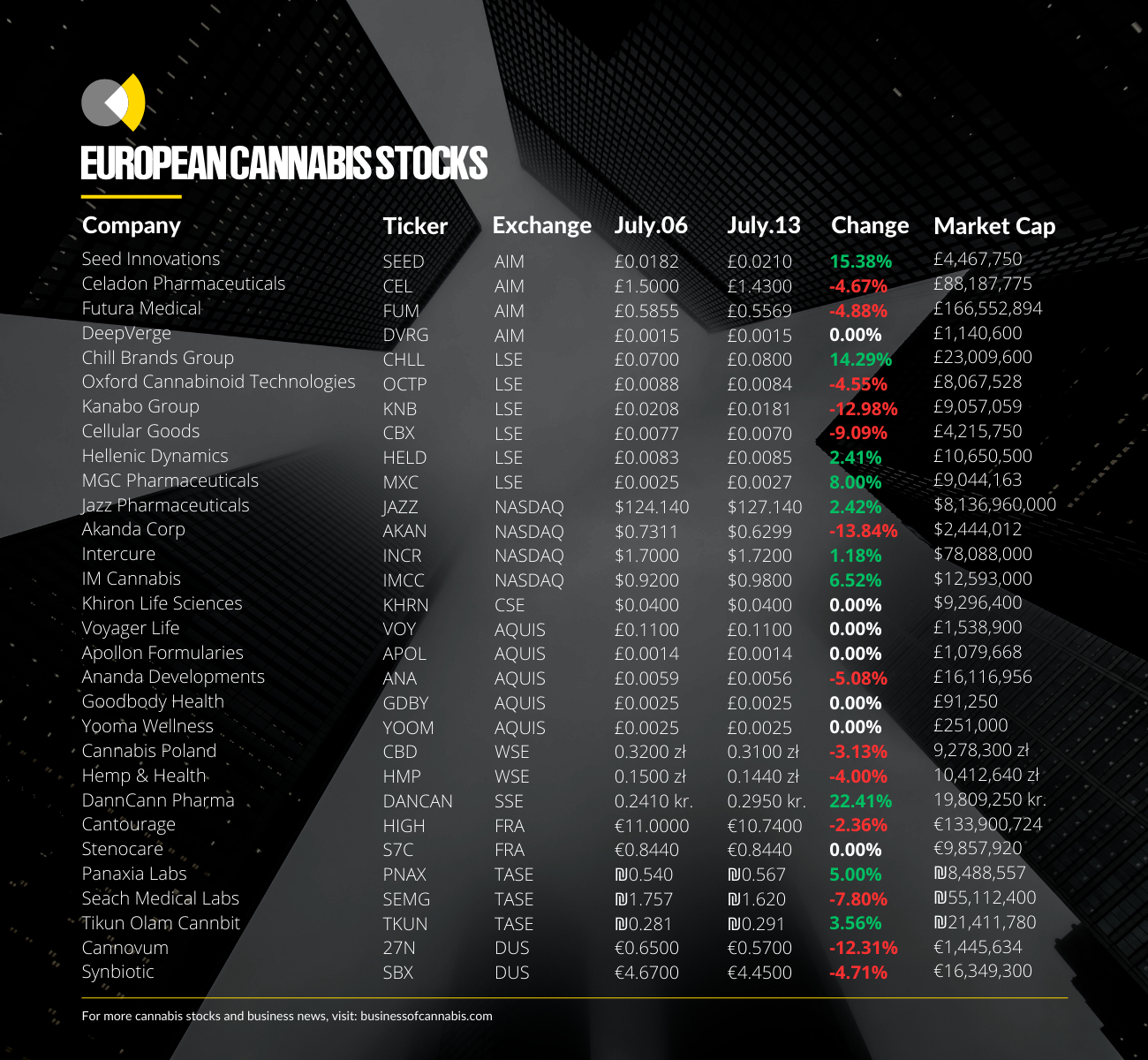
Cellular Goods
Cellular Goods has issued a long-awaited update to investors regarding its recent acquisition of carbon sequestration company King Tide Carbon (KTC).
On May 09, the CBD cosmetics company brand announced that it had closed the acquisition of KTC, satisfied by the issue of 95,000,000 new ordinary shares of £0.001 each in the company.
The unlikely acquisition left investors with a number of questions regarding the future direction of the company, and whether it signalled plans to further move away from its troubled CBD cosmetics range, which has struggled to gain sales traction since launch.
Investors had been assured of an update during the ‘second half of June’, but this came in the form of an interview with Proactive Investors and Cellular Goods ‘interim CEO’ Darcy Taylor.
According to Mr Taylor, the acquisition was part of the company’s ambition to ‘widen our lens of opportunity across the bio synthetics production fields’, announced during its recent interim results, allowing the company to ‘deepen its sustainability vision’.
He argued that despite the firm still being pre-revenue, the ‘unprecedented demand for innovation’ in carbon removal presented an opportunity for the company to leverage its ‘bio synthetics experience’.
“Although KTC is a pre-revenue, early-stage carbon removal startup, it’s now we’re entering that second phase of development prior to commercialisation. We anticipate that we’ll finalise multiple carbon removal projects in the next 12 months.”
Finally, Mr Taylor was asked about his position as interim CEO, given that he had now held the position for around a year.
He said that there was ‘no definitive deadline’ for his tenure, but that the company was keeping a ‘close eye’ out for potential candidates.
With the company having reported sales of just £31k in its recent interim results, pressure is growing on Mr Taylor, who remains the CEO of Pioneer Media Holdings, to generate revenues before Cellular Goods’ £2.98m in remaining cash runway is used up.
Little Green Pharma
Australian Stock Exchange (ASX) listed Little Green Pharma (LGP), which is also part of London Stock Exchange (LSE) listed SEED Innovations’ investment portfolio, saw its stock jump nearly 20% this week.
The stock bump came as LGP, which already supplies medical cannabis oil to the French pilot programme, announced that it had won a second call for tenders.
LGP will now supply up to US$1.6m (US$77 per bottle) of its CBD50 medical cannabis oil to the French pilot programme, which is due to end in March 2024.
According to the company, LGP remains the largest supplier of the experiment, seeing an estimated 85% of its 3,000 participants trial its products.
Newsweed reported in March that LGP, which has been supplying the trial since its inception three years ago, had initially refused to apply for the first call for tenders after the experiment was extended, seeing the French Ministry of Health offer to pay just €14 ($15.45) per bottle.
The poor response from companies to this initial tender call led to a number of supply shortages for patients, leading the government to issue this second call for tenders.
“The Company remains very buoyant about the potential of a future French medicinal cannabis industry and believes its success in the trial continues to confirm the advantages of developing a robust, export-led sales strategy from its assets in Australia and Denmark.”
MGC Pharmaceuticals
MGC Pharmaceuticals also saw an uptick in its stock price this week after receiving a second purchase order for its proprietary ArtemiC product.
On July 07, MGC announced that AMC Pharma had placed a second$1m purchase order for 100,000 units of ArtemiC.
This will consist of a $150,000 immediate down payment, with the remaining $850,000 set to be provided upon completion of production and dispatch of the order, which is expected to be completed by September 2023.
MGC and AMC’s new deal forms the latest extension of a long running partnership between the two companies, which signed a $24m multi-year ‘market supply and distribution’ agreement in August 2021.
CEO and Managing Director of MGC Pharmaceuticals Roby Zomer said: “This order from AMC validates ArtemiC for the US market and demonstrates our ability to supply an FDA-authorised product in a world-leading market. This order, combined with AMC’s new supply agreement to distribute across the US, gives us confidence for the future growth of MGC.”





















